Medical Packaging
Do you need precision medical blister packaging for your healthcare products? BlissPak delivers healthcare thermoformed packaging that meets critical safety and regulatory standards. In a market where reliability matters, our medical blister packaging ensures your products are protected and professionally presented. Designed to fit your exact specifications, whether for surgical instruments, implantables, unit-dose pharmaceuticals, or diagnostic devices. Fully customizable and engineered to provide secure sterile barriers. So request your free packaging assessment now!
Medical Packaging FAQs
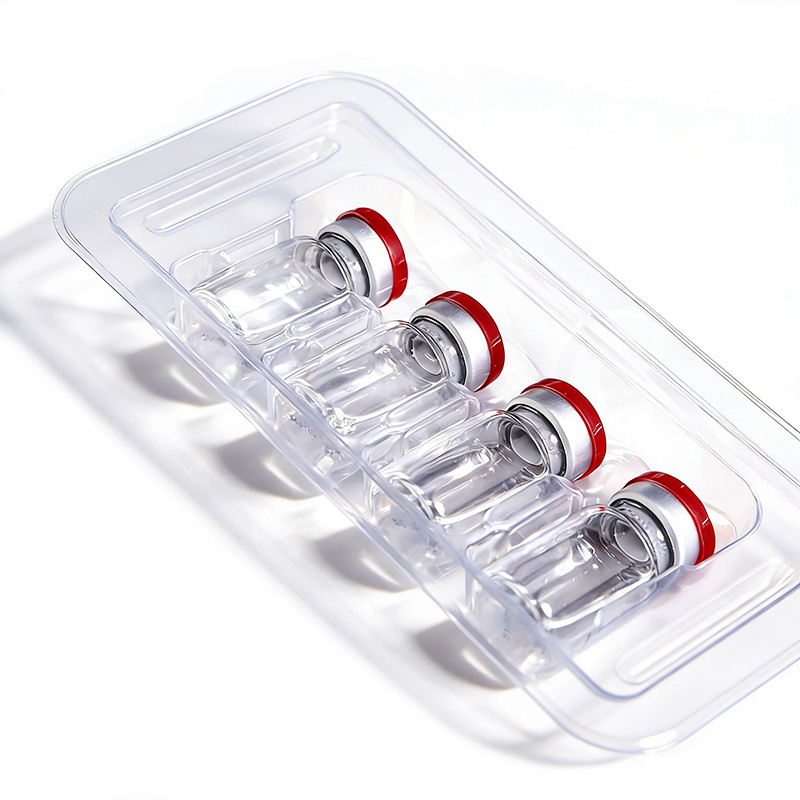
Customizable heat-sealable structures can be tailored to specific tray applications, paired with medical-grade dialysis paper to achieve sealed sterile protection. Alternatively, sterile individual outer packaging is available, where trays are placed into medical sterile pouches and vacuum-sealed to meet sterile requirements in open-use settings such as operating rooms and laboratory departments.
Medical packaging utilizes thickened medical-grade materials combined with an impact-resistant structural design. The blister edges feature collision-proof reinforcement, while heat-sealed edges incorporate double-layered thickening. After undergoing drop hammer impact and stacking compression tests, the packaging exhibits no seal cracking or blister damage under routine logistics compression, ensuring the sterile barrier remains unaffected.
Customizable medical-grade antistatic thermoformed plastic material meets medical biocompatibility requirements with no harmful substance leaching. Compatible with medical electronic devices such as patient monitor accessories, ultrasound probes, and ECG electrode pads, it provides both sterile protection and electrostatic shielding.
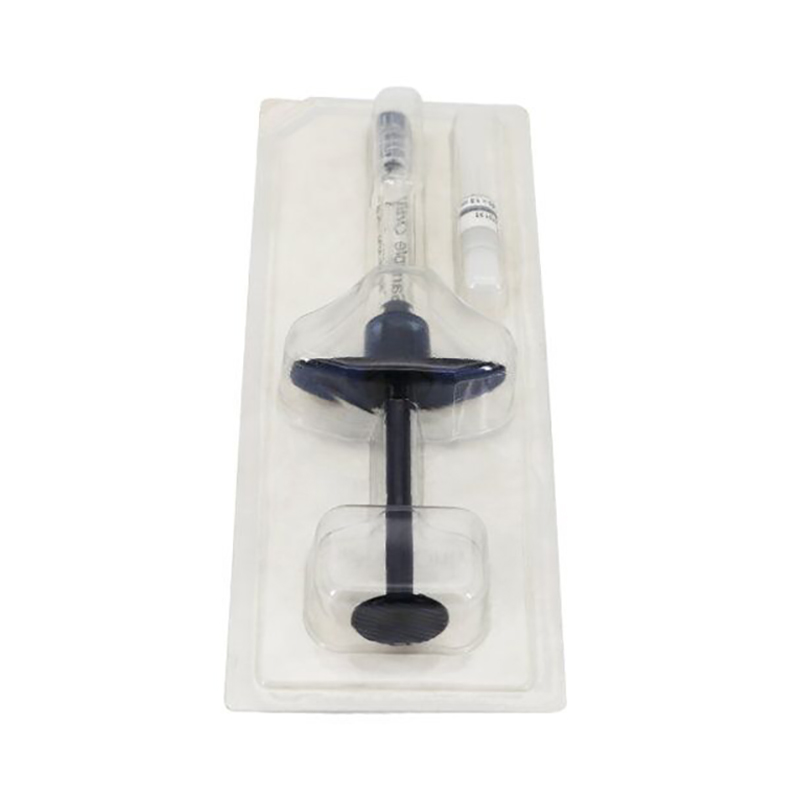
For reusable applications, customizable resealable structures can be provided. These feature medical-grade sterile self-sealing strips and lids, enabling resealing after opening to maintain sterility of remaining contents. Alternatively, individual small packages are available to prevent sterile contamination from opening the entire package.
If the customer detects that the product's sealing performance or sterility fails to meet standards upon receipt, they must provide feedback within 7 business days. We will respond within 24 hours and arrange for professional personnel to conduct verification testing. If it is confirmed to be a quality issue on our part, we will provide a full replacement at no cost and assume responsibility for any medical product losses resulting from packaging defects, ensuring full compliance with the customer's production requirements throughout the process.
You may want to know about Medical Packaging
Get sterile-safe, contamination-resistant medical blister packaging for your pharmaceuticals, surgical tools, diagnostic components and disposable medical supplies that meet global healthcare regulatory standards. There are many critical factors you need to consider when thinking about quality medical blister packaging. Putting your efforts into medical blister packaging can reinforce your brand’s reputation for safety and reliability among healthcare professionals and end-users.
Wholesale medical blister packaging with clear, compliant labeling streamlines clinical use and inventory management for pharmacies, hospitals and labs. Ensure that your blister packaging design focuses on precision-molded cavities and medical-grade materials to preserve sterility and prevent physical damage. For professional branding, add your brand logo, product specifications and sterility indicators to align with industry standards.
High-End Medical-Grade Materials for Medical Blister Packaging
The right medical blister packaging safeguards product integrity and builds trust in your healthcare brand. Choose certified, sterile-compatible blister materials for your medical products to prevent contamination, moisture damage and tampering during storage and shipping.
In more detail, small medical components like test strips and syringes require precision blister cavities with clear labeling to improve usability. Whereas larger surgical tools and diagnostic devices need heavy-duty medical blister trays to keep items securely organized and protected.
Custom Sizes for Every Medical ProductOne size never fits all in medical packaging. We provide fully customized medical blister packs built to your exact product dimensions.
Whether for tiny components, syringes, surgical tools, or portable devices, we create blister packaging with precise measurements to ensure a perfect, protective fit. Share your specifications, and we will deliver accurately sized blister packs tailored to your exact requirements.
Precision Design for Medical Safety & UsabilityMedical blister packaging is engineered for practicality, hygiene, and compliance. Each design is smooth, user-friendly, and built to support safe handling in hospitals, clinics, labs, and pharmacies.
Our functional medical blister styles include:
Cavity Blister Trays
Custom-molded cavities securely hold surgical tools, diagnostic parts, implants, and small medical devices. They prevent movement, scratches, and damage, while supporting organized storage and quick access.
Sealable Medical Blisters
Designed for tamper evidence and sterility, these blister packs ensure products remain safe and uncontaminated until use. Perfect for single-use medical instruments and pharmaceutical items.
Clamshell Medical Blisters
All-in-one protective design that encloses devices fully while allowing clear visibility. Sturdy, reusable, and ideal for larger or delicate medical tools that require ongoing protection.
Sterilization-Safe Blister Packs
Specially constructed to withstand standard sterilization processes while preserving product integrity. Essential for surgical and clinical-grade medical supplies.
Medical packaging should reflect precision, cleanliness, and professionalism. We offer refined finishes and branding solutions that elevate your image while complying with industry standards.
Our customizable branding and finishing options include:
- Clean, crisp logo printing
- Product labeling and usage instructions
- Anti-fog and anti-smudge finishes
- Sterility indicators and compliance markings
- Tamper-evident sealing solutions
Each detail enhances usability, professionalism, and brand recognition in medical and pharmaceutical markets.
Order Your Custom Medical Blister Packaging Today
From production to delivery, we maintain strict quality at every step. Each order is carefully checked before shipment to ensure there are no defects or inconsistencies. We deliver reliable, high-quality medical blister packaging efficiently, with consistent lead times and dependable service. Our goal is to provide smooth, stress-free experiences for brands that value safety and professionalism.
If you’re looking for consistent, safe, and customizable blister packaging for medical products, we’re here to help. Get in touch with us to learn more about our solutions and find the right packaging for your needs.